|
Self-healing rubber is an example of a molecular solid with the potential for significant commercial applications. Thus toluene (C 6H 5CH 3) and m-xylene have melting points of −95☌ and −48☌, respectively, which are significantly lower than the melting point of the lighter but more symmetrical analog, benzene. If the molecules have shapes that cannot pack together efficiently in the crystal, however, then the melting points and the enthalpies of fusion tend to be unexpectedly low because the molecules are unable to arrange themselves to optimize intermolecular interactions. The enthalpies of fusion also increase smoothly within the series: benzene (9.95 kJ/mol) < naphthalene (19.1 kJ/mol) < anthracene (28.8 kJ/mol). For example, the melting points of benzene (C 6H 6), naphthalene (C 10H 8), and anthracene (C 14H 10), with one, two, and three fused aromatic rings, are 5.5☌, 80.2☌, and 215☌, respectively. For similar substances, the strength of the London dispersion forces increases smoothly with increasing molecular mass. 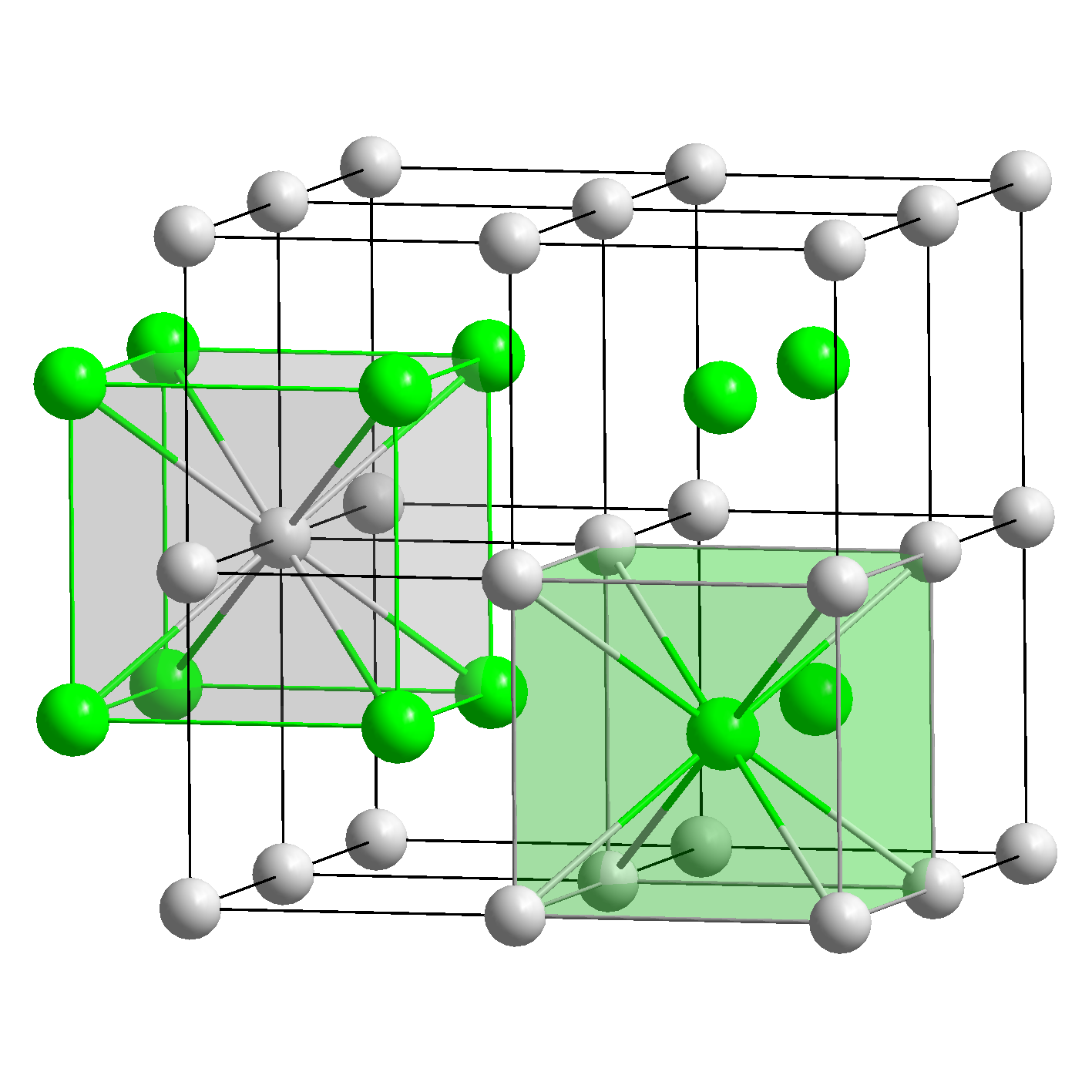 
In solid benzene, the molecules are not arranged with their planes parallel to one another but at 90° angles.īecause the intermolecular interactions in a molecular solid are relatively weak compared with ionic and covalent bonds, molecular solids tend to be soft, low melting, and easily vaporized (Δ H fus and Δ H vap are low). In both cases, however, the values are large that is, simple ionic compounds have high melting points and are relatively hard (and brittle) solids. The forces that hold Ca and O together in CaO are much stronger than those that hold Na and F together in NaF, so the heat of fusion of CaO is almost twice that of NaF (59 kJ/mol versus 33.4 kJ/mol), and the melting point of CaO is 2927☌ versus 996☌ for NaF. Because of the higher charge on the ions in CaO, however, the lattice energy of CaO is almost four times greater than that of NaF (3401 kJ/mol versus 923 kJ/mol). 
For example, NaF and CaO both crystallize in the face-centered cubic (fcc) sodium chloride structure, and the sizes of their component ions are about the same: Na + (102 pm) versus Ca 2+ (100 pm), and F − (133 pm) versus O 2− (140 pm). The lattice energy, the energy required to separate 1 mol of a crystalline ionic solid into its component ions in the gas phase, is directly proportional to the product of the ionic charges and inversely proportional to the sum of the radii of the ions. (For more information about ionic solids, see Section 4.2.) The strength of the attractive forces depends on the charge and size of the ions that compose the lattice and determines many of the physical properties of the crystal. consists of positively and negatively charged ions held together by electrostatic forces. You learned in Chapter 4 that an ionic solid A solid that consists of positively and negatively charged ions held together by electrostatic forces.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
